 What is the relationship between what you know and what you need to find out? What information have you been given in the question? 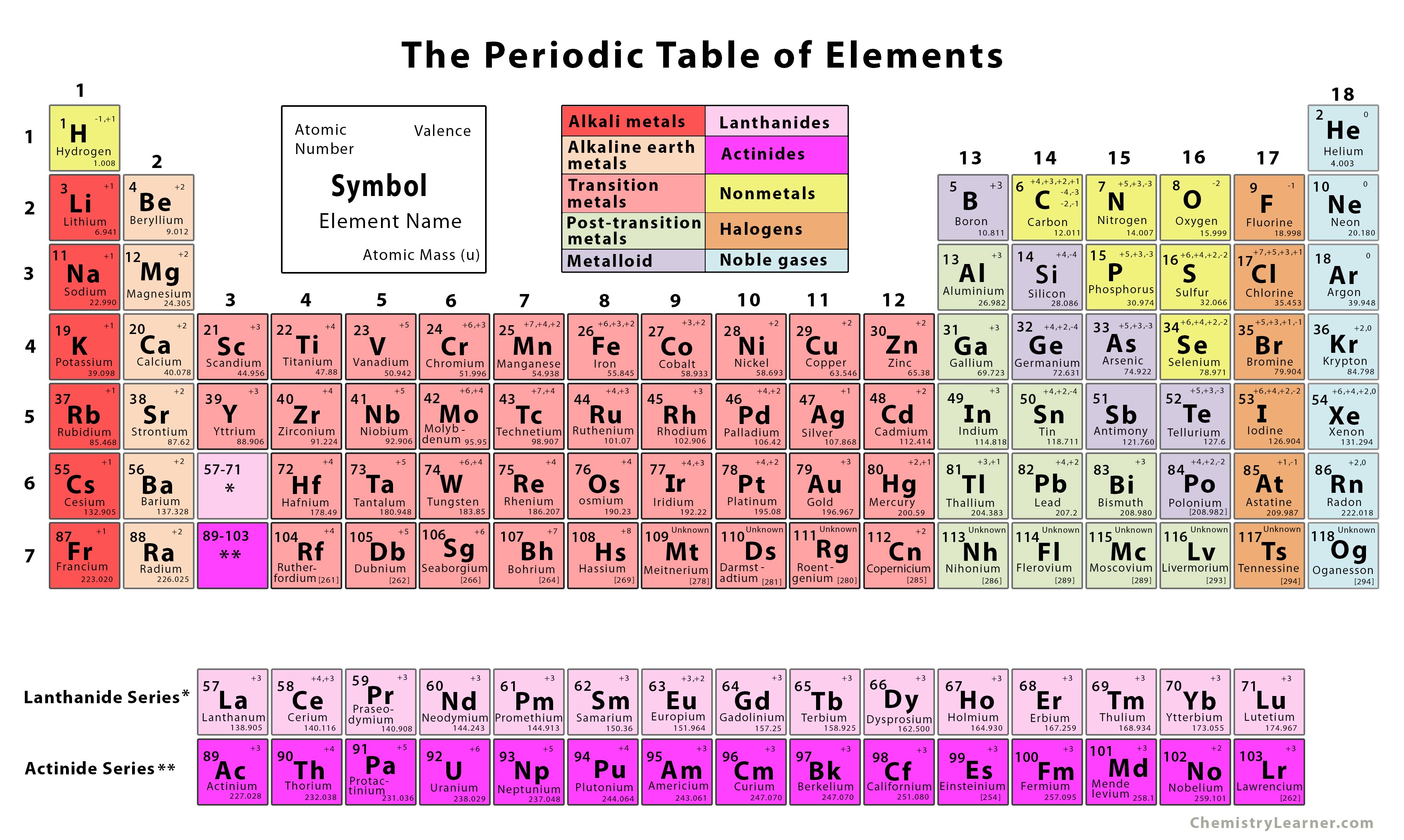
Oxygen gas (chemical formula O 2) makes up about 21% of the Earth's atmosphere.ĭecide whether oxygen gas is an element or a compound. The most common elements found in living things are: oxygen (which makes up about 21% of the atmosphere)Ĭommon elements found in the Earth's crust are:.nitrogen (which makes up about 78% of the atmosphere).The Earth's atmosphere is made up of a mixture of different gases, but is mostly made up of two elements: The most common elements found in the universe are: Neon has is an element with chemical symbol NeĬhemistry teachers often ask their students to memorise the names and chemical symbols of the first 20 elements in the periodic table of the elements, so here they are: (2) Helium is an element with the chemical symbol HeĬalcium is an element with the chemical symbol Ca ⚛ if there is a second letter in the chemical symbol of an element it is always a lower case letter Nitrogen is an element with the chemical symbol N Hydrogen is an element with the chemical symbol HĬarbon is an element with the chemical symbol C ⚛ the first letter of the chemical symbol of an element is always a capital letter (upper case letter) The chemical symbol of an element can be made up of either 1 letter or 2 letters: This chemical symbol of an element is most often based on the current name of the element, or, in some cases, it is based on an older name for the element. In addition, there are more than 20 man-made elements (synthetic elements or artifical elements).Īll the known elements, both naturally occurring and synthetic elements, are listed in the Periodic Table of the Elements.Įach element has been given a unique name and a unique chemical symbol. There are 92 naturally occurring elements. View Notes.An element is defined as a pure substance that cannot be decomposed (broken down) into simpler substances.įor example, if you heat nitrogen gas it will get hotter, but it won't break down (decompose) into anything simpler than nitrogen. Technical information, teaching suggestions, and related resources that complement this Concept Builder are provided on the Notes page. Learners and Instructors may also be interested in viewing the accompanying Notes page. However, the 12 different groups of questions can be printed. There is no need for an activity sheet for this Concept Builder. Users are encouraged to open the Concept Builder and explore. 
The built-in score-keeping makes this Concept Builder a perfect candidate for a classroom activity. They are organized into 12 different Question Groups and spread across the three difficulty levels. Question-specific help is provided for each of the 12 situations. There are three levels of difficulty with each successive level including more questions and more difficult questions.There are a total of 24 questions in this Concept Builder. Learners should build a comfort with the use of terms like period, group, atomic number, atomic mass, metals, nonmetals, metalloids, transition metals, alkali metals, alkaline earth metals, halogens, and noble gas. The Name That Element Concept Builder provides learners an exercise in understanding the organization of the elements on the Periodic Table. Concept Builders » Chemistry » Name That Element 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
